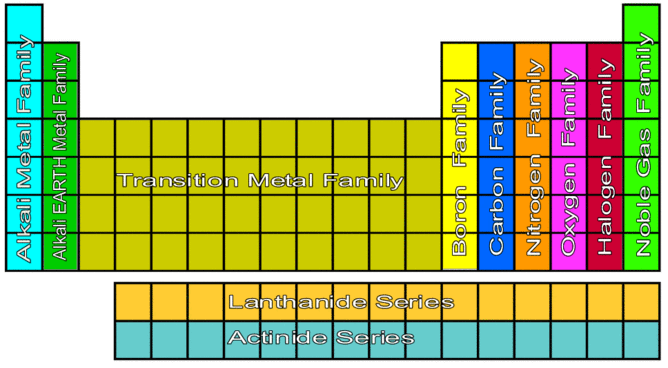
The Groups:
Alkali Metals:
The Alkali Metals are the first group on the periodic table. Here are some facts about these metals
- Have one valence electron.
- Extremely reactive.
- Never found in nature in their pure form.
- Silver colored and shiny
- Low enough density to be cut by a knife
- Generally stored in oil
- Cesium and rubidium are used to make atomic clocks
- Form salts by combining with halogens
- Sodium and potassium play an important biological role for life on Earth and we cannot live without them.
Alkaline-earth metals:
Alkaline-earth metals are the second group on the periodic table. Here are some facts about these metals:
- Reactive, but slightly less so than alkali metals
- Silver colored
- Denser than alkali metals
- Have 2 valence electrons
- All occur in nature, but are only found as compounds and minerals (not in elemental form)
- All tend to form ionic bonds (except beryllium, who forms covalent bonds)
- Burn with various color flames (beryllium- white, magnesium- bright white, calcium- red, strontium- crimson, barium-green, and radium-red)
- Calcium helps build strong bones and magnesium is used to regulate the body's temperature
- Radium is formed from decaying uranium, causing it to be radioactive and dangerous to handle
- Magnesium and calcium also have many uses in manufacturing and industry
Transition Metals:
Transition metals are all the elements listed in groups 3-12. Here are some characteristics of transition metals:
- Shiny and good conductors of heat and electricity
- Higher densities than alkali and alkaline-earth metals
- Variable valence electrons
- Paramagnetic
- Conduct electricity
- Transition metals are call the "d-block" on the periodic table, and there are 35 elements located here.
- Iron, cobalt, and nickel are the only three elements that produce a magnetic field.
- Transition metals are used a lot in industry for various reactions because of their unique qualities.
Lanthanides and Actinides:
Lanthanides and Actinides are transition metals as well, but they are placed at the bottom of the table (to keep the table more compact). Here are some properties are lanthanides and actinides:
- Lanthanides are shiny and reactive.
- Actinides are all radioactive and unstable.
- Elements 95-103 are not found in nature, but instead created in a lab.
- Uranium and plutonium are used in nuclear reactors and nuclear bombs.
- Lanthanides are used in hybrid cars, superconductors, and permanent magnets.
- Actinide americium is used in smoke detectors.
- All of the lanthanides have at least one stable isotope, except for promethium.
- None of the actinides have a stable isotope (all radioactive).
Boron Group:
Group 13 is known as the Boron Group. Here are some characteristics of the Boron family:
- Have 3 valence electrons
- Contains 1 metalloid (Boron)
- Contains 4 reactive metals, which are solid metal under standard conditions
- The 4 reactive metals are also malleable, ductile, and good conductors of electricity and heat
- The 4 reactive metals have a high density and are softer and have lower melting and boiling points than transition metals
- Aluminium is the most abundant metal in earth's crust and the third most abundant element.
- Thallium is highly toxic and can cause death.
- Gallium's melting point is slightly higher than room temperature, so it will melt if held in the hand.
Carbon Group:
The Carbon group is group 4a on the periodic table. This group has:
- 4 valence electrons
- varied reactivity
- a nonmetal (C), two metalloids (Si, Ge), and 2 metals (Sn, Pb).
Nitrogen Group:
The Nitrogen Group is group 5a on the periodic table. This group has:
- 2 nonmetals (N, P), 2 metalloids (As, Sb), and 1 metal (Bi).
- varied reactivity
- 5 valence electrons
Oxygen Group:
The Oxygen Group is group 6a on the periodic table. This group has:
- 3 nonmetals (O, S, Se), 1 metalloid (Te), and 1 metal (Po).
- very reactive
- 6 valence electrons
- Nitrogen makes up 78% of Earth's atomsphere, white oxygen makes up around 21%.
- Only 18 elements on the periodic table are considered nonmetals.
- Carbon is the most important element to the existence of life on Earth.
Halogens:
Halogens are group 7a on the periodic table. They are located to the right of other nonmetals and to the left of noble gases. They have many unique properties, including the following:
- Have 7 valence electrons
- Fairly toxic
- Form acids when combined with hydrogen
- Highly reactive and electronegative
- All nonmetals
- Poor conductors of heat and electricity
- Tend to form salts with metals (ex: NaCl- table salt)
- Fluorine is one of the most reactive elements in existence.
- Fluorine gas is deadly.
- Astatine is used in medicine.
- Bromine liquid evaporates at room temperature and emits an orange vapor.
- Even though fluorine and chlorine are poisonous, they are essential to human health.
Noble Gases:
Nobles Gases make up group 8a. They all share some common characteristics, including:
- Unreactive metals
- Colorless, odorless gases at room temperature
- All found in Earth's atmosphere is small amounts
- Have 8 valence electrons
- Helium is the second most abundant element in the universe after hydrogen.
- Helium in non-flammable, so it is safer to use in balloons than hydrogen.
- Helium has the lowest melting and boiling points of any substance.
- Neon signs use neon gas as well as a mixture of noble gases to create the bright lights and various colors.
For more information on the groups of the periodic table, visit www.ducksters.com/science/chemistry/